

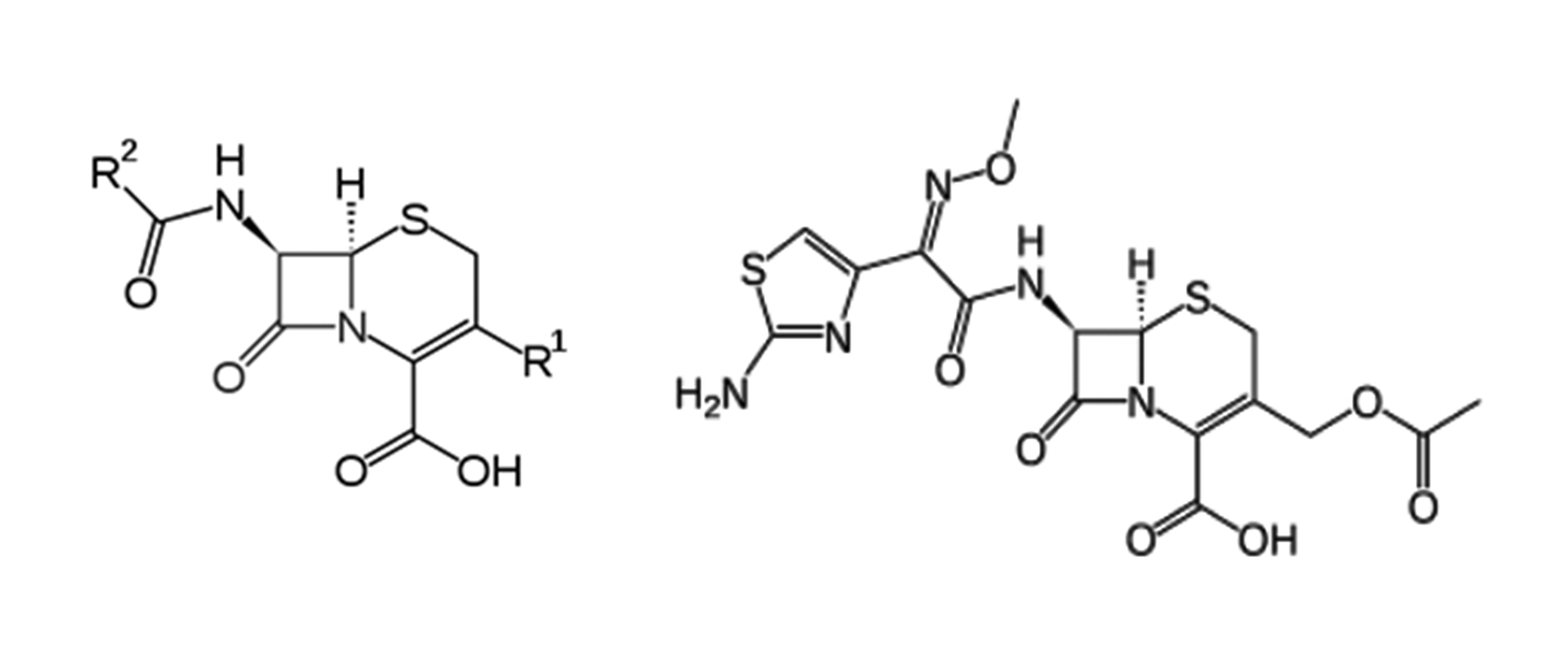
Cefotaxime (Cfm) is a member of the third generation of the Cephalosporin antibiotics. It used on a wide scale in prescribed antibiotic drugs as anti-infection for gram-positive microorganisms and gram-negative microorganisms. The present study aimed to develop an HPLC method of Cfm analysis enjoyed highly linearity, repeatability, robustness, ruggedness, selectivity, rapidly and economical to use. The chromatographic method uses a reversed phase column BDS column (150 mm x 4.0 mm x 5 μm). The mobile phase was prepared by mixing Methanol: Phosphate buffer (1000 mL : 130 mL) and the pH was adjusted to 6.15 at isocratic flow rate 1.0 mL/min with PDA detector at 235nm, column oven adjusted at 30°C and injection volume 20 μL. The method revealed that satisfied linearity regression R2 (0.9992) with repeatability (0.15%) with DL and QL; 35.5 ng/mL and 107.6 ng/mL respectively. The method showed a successful application of analytical method validation for Cfm in bulk and pharmaceutical formulations.

Scheduled Server Maintenance and System Downtime Notice Dec 16, 2025
Celebrating CM Editorial Board Members Recognized in the Wor... Oct 10, 2025

Food Science and Engineering Now Indexed in CAS Database Aug 20, 2025

Contemporary Mathematics Achieves Significant Milestone in 2... Jun 19, 2025

Three Journals under Universal Wiser Publisher are Newly Ind... Apr 21, 2025